IOTM: Suprachoroidal ILUVIEN® Implant for DME
Image of the Month (IOTM) is a collection of interesting clinical cases with high quality images for all relevant imaging modalities (ex: color fundus, OCT, OCTA, FAF, FA, En Face, Red-free, choroidal vasculography (CVG), anterior imaging) and other clinical results if relevant (ex: visual field plots). Each case is submitted by an eye care professional using one of Topcon’s industry-leading OCT devices.
Case background
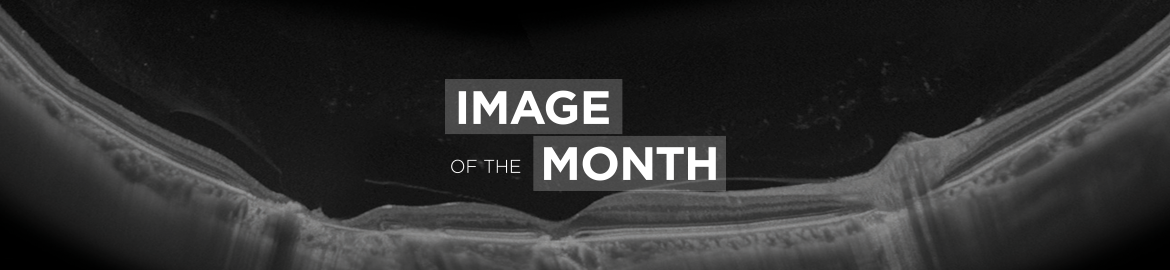
This is a patient with Chronic Diabetic Macular Edema. The patient received several anti VEGF injections; the need of reinjection was a problem. The patient was treated by a suprachoroidal ILUVIEN® implant. The implant is a slow release device that releases f luocinolone acetonide at an initial rate of 0.25 µg/day (average rate 0.2 µg/day) and lasts 36 months. The implant is designed for intravitreal injection. We implanted the device in the suprachoroidal space in part of a trial to see if this would have an effect on controlling the macular edema, and at the same time minimizing or preventing the long term effect of steroids on the eye seen from intravitreal exposure as Cataract and Glaucoma. The trial was based on our previous studies that showed the therapeutic effect of suprachoroidal Triamcinolone efficacy. The implant was placed in the suprachoroidal space using a sclerotomy created 4mm behind the limbus. The implant was placed in a special designed catheter to inject it in the suprachoroidal space below the disk with the releasable end towards the macula. Color photography as well as OCT show and confirm the suprachoroidal placement of the implant.
—Ehab El Rayes, MD, PhD
Diagnosis: Chronic Diabetic Macular Edema
Captured with: Topcon DRI OCT Triton
DRI OCT Triton Images:
A. B. Color Fundus Image Swept Source OCT
B-scan Image
C–F. OCT Angiography Images
OCT Angiography is not available for sale in the US.
The opinions, ideas, views and assumptions expressed are the author’s own and do not necessarily represent the views of Topcon, nor do they constitute advice from Topcon.