The Importance of Image Quality in Accurate Diagnosis
Imaging technologies play an important role in our profession, and the ability to visualize tissue and evidence of disease in detail is one of the cornerstones of our jobs. High-quality images are critical to reaching a more confident diagnosis, and ultimately, delivering better patient outcomes through more informed disease management. At the eyeRISE 2021 virtual conference, I discussed “Advancements in Optical Coherence Tomography (OCT) Imaging Devices” with several of my colleagues. We agreed that image quality is necessary for accurate interpretation and analysis, yet there still are challenges to yielding such optimized images.
When considering OCT imaging devices, the clinical usefulness of a scan can be affected by three parameters: 1) the scan area (field of view); 2) the scan density (resolution); and 3) the scan time for image acquisition. If we hold any one of these parameters constant, the other two factors can be inversely affected. For example, if we want a fast scan acquisition with high resolution then we are limited in scan area. Inherently, there has always been a need to make some tradeoff when selecting the scan pattern on our OCTs—until now.
In an effort to eliminate the need for eye care providers to have to choose between scan area and scan density, Topcon Healthcare (Tokyo, Japan) developed PixelSmart™ technology for the DRI OCT Triton, Topcon’s Swept Source OCT (SS-OCT) platform. At its core, PixelSmart is designed to deliver the best of both worlds—the image quality of a high-density line scan and the wide coverage of a dense cube scan—without sacrificing scan speed.
PixelSmart’s new image processing algorithm is elevating visualization of the retina by delivering the clarity of averaged images throughout the entire volume scan—reducing speckle noise and improving contrast. The technology is a post-processing technique, meaning scan time is not affected, and Triton scans previously captured on the device can be reprocessed to further enhance scan quality.
This step forward in OCT imaging aims to provide clinicians with the highest possible image quality to help them better identify and differentiate between pathologies, with the goal of improving patient care and outcomes.
In the following discussion, I share my first impressions with PixelSmart technology after evaluating it in my clinic.
OCT in Clinical Practice
What is your typical imaging protocol in clinic?
Dr. Haynie: Our office protocol entails volumetric scans on all new patients. In addition, if we are dealing with a patient with age-related macular degeneration, we generally use high-resolution scan patterns to get a better view of potential neovascular membranes and subretinal fluid. For our diabetic patients or those with retinal vascular disease, we rely primarily on volumetric scans.
What percentage of your patients have cataracts or other media opacities and how is SS-OCT technology impacting your care of these patients?
Dr. Haynie: About 30 percent of our patients have significant media opacities. When you are trying to diagnose, manage and stabilize retinal disease prior to cataract surgery, it can be challenging to evaluate whether a patient is ready to go forward with the procedure. For that reason, we use SS-OCT technology to penetrate through media opacities and help us make that assessment.
When assessing patient findings, why is it still important to look through the B-scans for every patient rather than just reviewing OCT reports?
Dr. Haynie: I think one of the upsides of OCT technology is the algorithmic data it gives us, but that can also be a downside with regard to, for example, “red disease.” You can look at a thickness map and see a large area of increased retinal thickening—it shows up as red areas on these reports—but you really don’t know what has caused that. So, just like when you see a lesion on a fundus photo, you need to look at the live tissue. The problem with relying on thickness maps alone becomes apparent when a patient has a retinal cotton-wool spot or a significant intraretinal hemorrhage. Those conditions will create a very large area of increased retinal thickness. However, cotton-wool spots can improve over time, so it’s important to go through the B-scans to try to isolate and identify the origin of the pathology that has resulted in elevation or thinning on the individual retinal thickness map.
How has PixelSmart helped to optimize your clinical workflow?
Dr. Haynie: Most technologies today offer faster and faster scans, so speed is readily available. But without PixelSmart technology, interpreting the raw data and multiple scans can be challenging. With PixelSmart, you get speed, large volumetric data scans, and high-quality images. As a result, you have the information you need from one scan rather than having to take that patient through multiple scans, such as raster scans, 5-line scans, and radial scans. That is extremely helpful in optimizing clinic workflow because we’re inundated with retinal disease cases. Every patient coming in is getting OCT imaging, but now we only need one scan to get all of the information we need.
Applying PixelSmart to Patient Cases
CASE STUDY 1: Central Serous Chorioretinopathy: PixelSmart Enhances the View of the Choroid
A 74-year-old woman was referred to my clinic for management of Central Serous Chorioretinopathy (CSC) in the right eye. She described a gray smudge in the central vision of her right eye that appeared to have grown larger over the previous six weeks. She had a medical history of hypertension and her ocular history was unremarkable. When thinking about the typical demographic of patients with CSC, this patient was a little bit older than we typically see, and her visual symptoms raised suspicion as to whether this was the correct diagnosis.
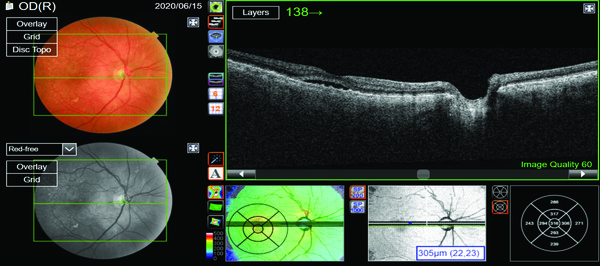
Looking at the original B-scan of the patient’s right eye in Figure 1, there is a presumed neurosensory retinal detachment, which is the shallow pocket of subretinal fluid.
When PixelSmart is toggled on as shown in Figure 2, we can view the high-resolution neurosensory detachment and choroidal scleral junction. Diseases of the pachychoroid, which include CSC, don’t tend to exhibit this razor-thin choroid as in our patient. Yet, PixelSmart reveals thinning of the choroid indicating that a closer look is necessary to identify the source of the subretinal fluid.
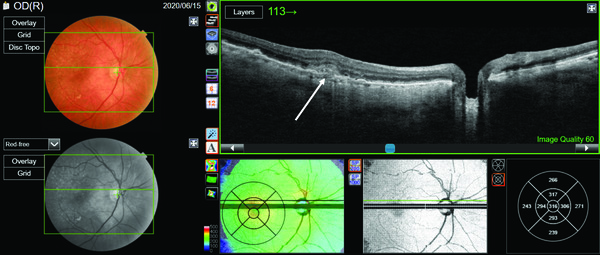
In this case, it is necessary to scroll through the OCT B-scans starting with the superior B-scan (Figure 3) just above the fovea. We now see an area of RPE disturbance, a break in Bruch’s membrane, subretinal thickening as well as the presence of a shallow, irregular retinal pigment epithelial detachment known as the “double-layer sign.” These are all indications of a choroidal neovascular membrane (CNV) that has migrated through Bruch’s membrane and is growing into the subretinal space.
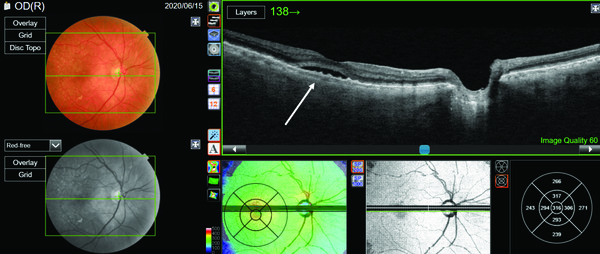
In this case, the utilization of PixelSmart helped us to differentiate between CSC and CNV, which is critical as the etiology, management, and the long-term prognosis of the two conditions is quite different. The patient went on to receive serial anti-VEGF injections and has done very well.
CASE STUDY 2: Lamellar Macular Hole or MacTel? PixelSmart Helps Make the Diagnosis
A 75-year-old woman was referred to our clinic for management of bilateral lamellar macular holes. Her chief complaint was that she was missing letters while reading and recently noticed areas of central distortion in each eye. The patient’s medical history was unremarkable and her ocular history revealed she was pseudophakic, having undergone cataract surgery three years prior.
Looking at the patient’s standard imaging in Figure 1, the B-scan through the central foveal area reveals a cavitation defect within the inner retina. This led to the initial diagnosis of a lamellar macular hole. However, on closer evaluation of the areas surrounding the cavitation defect, nothing stands out as being abnormal.
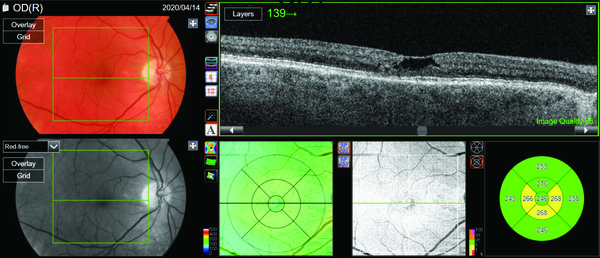
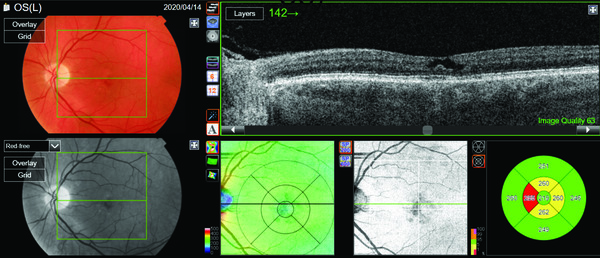
Standard imaging of the fellow eye in Figure 2 reveals a similar image, with a cavitation defect within the inner retina with adjacent structures and the RPE appearing intact.
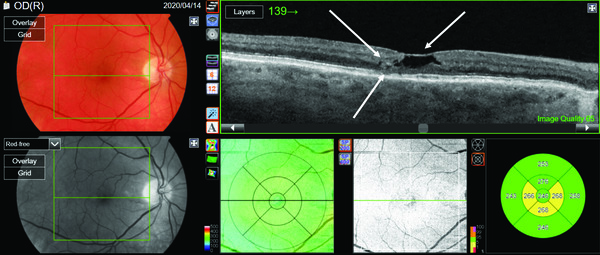
Toggling on PixelSmart in Figure 3 to view the initial line scan of the right eye, we see a cavitation defect that looks very similar to the original OCT scan. However, just to the left of the cavitation, a hyperrefl ective lesion within the deep retinal layer is apparent. More importantly, the ellipsoid zone and the IPL layer are disrupted in the temporal perifoveal region.
Imaging of the fellow eye using PixelSmart in Figure 4 highlights the cavitation defect in addition to the disruption of the outer retinal complex and a drape of the internal limiting membrane on the surface, which are characteristic features of Type 2 Macular Telangiectasia (MacTel). Because of this technology, we moved from an initial diagnosis of a lamellar macular hole to the appropriate diagnosis, which is Type 2 Macular Telangiectasia.
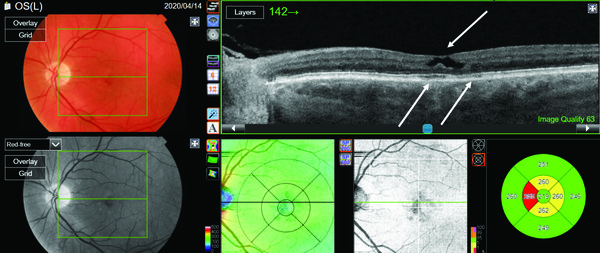
This differential diagnosis is particularly important because a lamellar macular hole typically carries a fairly good prognosis: it is not amenable to surgery, patients generally have stable vision, and the condition rarely progresses to a full thickness hole. Type 2 Macular Telangiectasia has a far different prognosis; patients may develop visual symptoms (as this patient did) but also serious complications, such as choroidal neovascular membranes, can occur.
As these cases demonstrate, high-quality imaging was necessary to make the more accurate diagnoses and determine the appropriate treatment plan for the patient. Innovations like PixelSmart are game-changing technology that bring immediate value to a clinical practice.

ABOUT THE AUTHOR
Maria Liu is an Associate Professor at Pacific University College of Optometry, and is the founder of UC Berkeley’s Myopia Control Clinic, the first of such nationwide. She received her bachelor’s degree of Clinical Medicine from Peking University, her OD from Pacific University, and her PhD and MPH from UC Berkeley. She is a world-renowned clinical researcher in the field of myopia and her focus is on the impact of complex multifocal environments on emmetropization and myopia development, as well as novel optical and pharmaceutical treatments in myopia retardation.